Exotic atoms may lead to new physics
Natural atoms only contain three kinds of subatomic particles: protons, neutrons, and electrons, but physicists are now creating exotic…
Natural atoms only contain three kinds of subatomic particles: protons, neutrons, and electrons, but physicists are now creating exotic atoms using other kinds of subatomic particles to learn more about how they work.
We tend to think of atoms as like little billiard balls of different kinds, but they are more like little machines. Each one works a little differently. To understand better how they work, it helps to replace one part with another to see what changes. Doing so, researchers have discovered that sometimes traditional models of how atoms work are wrong.
Indirect studies of exotic atoms go back to the 1950s with early synchrotrons and helium bubble chambers. Measurements from these experiments were very imprecise and could only confirm their existence but gradually methods improved and led to increasingly accurate experiments with a variety of atoms that cannot exist for long.
So far kaon, pion, muon, antiproton, and sigma hyperon atoms have all been observed using X-rays. The technique, called X-ray fluorescent spectroscopy, allowed measurements of state transitions in the atoms but direct synthesis and detection in order to do laser spectroscopy, which is much more precise, has been difficult.
In 2010, Randolf Pohl and others at the Max Planck Institute of Quantum Optics (MPQ) in Garching, Germany created muonic hydrogen where a muon replaces the electron. Using lasers they were able to calculate a charge radius of the proton which challenged known values. Muons are leptons like electrons with the same -1 charge and spin 1/2 and have no substructure, so creating muonic atoms isn’t too difficult.
Measurements of these exotic atoms enable us to study many aspects of atomic physics and verify predictions of the standard model to high precision. Strong interaction physics is a major motivation here and opens up our understanding of interaction strengths, masses, magnetic moments, nuclear properties of atoms, radioactive decay and capture, particle-nucleus interaction, and much more in basic science as well as having applications to nuclear fusion.
Recently, physicists have created even more exotic atoms from pions, which are mesons consisting of a quark and anti-quark. This is much more difficult because pions create a lot of new particles when you fire a beam of them at matter. To overcome that problem, researchers produced pions from graphite and directed the beam at ultra-cold superfluid Helium-4, creating pionic Helium!
Mesons like pions were predicted in 1935 following discovery of the neutron. The particle was predicted to be 200 times more massive than an electron. In 1947, charged pions were first detected, with about the correct mass, from cosmic rays. And in that same year two giants of atomic physics, Fermi and Teller, both predicted that exotic atoms could be formed by replacing the electron with the heavy, negatively charged meson.
Isolated, a pion decays into a muon and an anti-neutrino in only 26 nanoseconds. If you consider that a nanosecond is to a second what a second is to 32 years, that is pretty fast. So, that makes is really hard to study, but that’s nothing compared to what happens when you try to make an atom with a pion. In that case, the atom decays in less than a picosecond (a trillionth of a second) and the pion is absorbed into the nucleus. But, if placed into a stable configuration in a Helium atom, the pionic Helium is “metastable” meaning that it lasts much longer (10s of nanoseconds) but not indefinitely like ordinary Helium. The other electron (since, remember, Helium has two electrons and two protons) keeps the atom stable for longer even as the atom collides with other Helium atoms.
That longer lifetime makes pionic Helium suitable for laser spectroscopy using sub-nanosecond laser pulses which enables scientists to peer into the properties of this strange atom. The laser initiates a fission process, causing an electromagnetic cascade that sends the pion crashing into the nucleus like a spaceship shot down from orbit, and the fission out-products: neutron, proton, and deuteron, indicate that the laser hit a pionic Helium atom.
This technique could result in determining precise masses of pions as well as the muon antineutrino by comparing them with theoretical results from Quantum Electrodynamics (QED). Limits on new “fifth” forces can also be placed based on these measurement, constraining new physics.
This all comes down to the magic of spectroscopy, the same technique that we use to probe distant stars.
Spectroscopy allows scientists to study atomic transitions that occur at various frequencies of light. You can imagine an atom as being a little machine with a nucleus sitting at the center and electrons (or muons or pions) orbiting around it, separate from the nucleus but drawn to it by the electromagnetic attraction. Because atoms are so small, the electrons are delocalised, meaning that they appear in a probability cloud around the atom rather than like a little spaceship. Another feature of quantum mechanics is that the electrons can only live at precise levels of energy. They can have a “ground” state where they occupy the lowest level and they can have higher states. Atoms with multiple electrons can only have a certain number in each level.
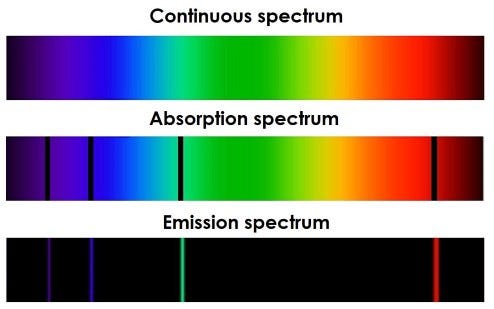
Atoms can absorb light particles called photons only at frequencies that match the levels the electrons are allowed to occupy. Otherwise, they don’t absorb them at all. Those frequencies are part of the atom’s spectrum. When light of the correct frequency strikes the atom, the electron absorbs the energy and becomes excited. Later on it will decay back down to its ground state and release the energy as light of a particular frequency. Different elements have different spectra of light that they absorb. This is how we know what stars are made of. We can see their absorption spectra where the atoms inside them have absorbed some of the light.
Some pictures show this process as the photon impacting the electron and causing it to “boost” to the next level like a rocket accelerating.
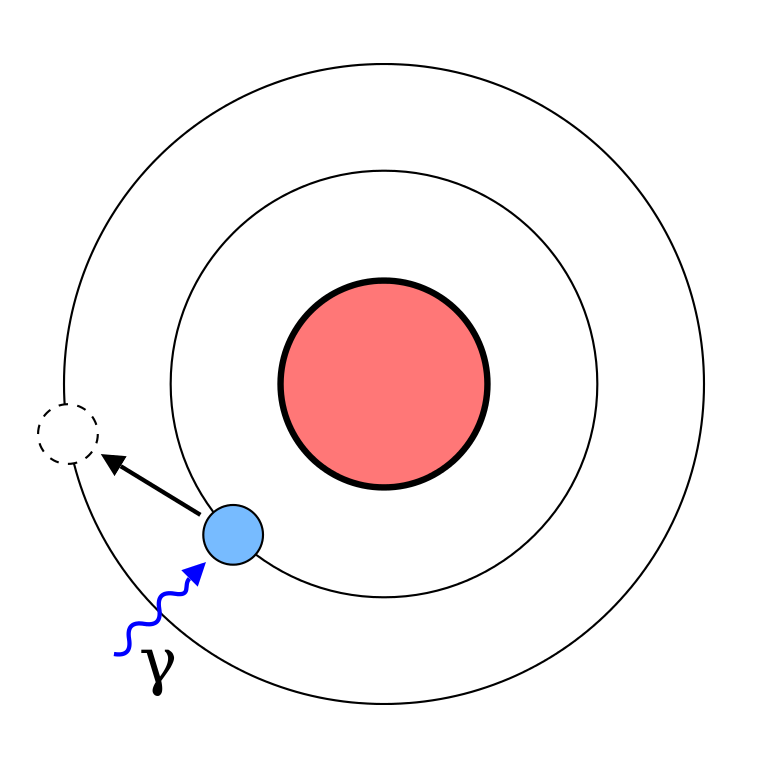
This picture is useful but wrong. The process is must more quantum mysterious than that. The electron cloud simply absorbs the energy and jumps to the next level. Without getting into the math, this is a better picture:
The photon causes the electron to jump instantaneously to a higher level.
An emission spectrum is the opposite of an absorption. In this case an atom has been excited in some way and then releases that energy as light at specific frequencies. This is how lasers are created.
Almost everything we measure with laser spectroscopy involves ordinary atoms, not pionic atoms, and the energy levels are well-known based on precise measurements and theoretical predictions of electrons. Pions, by contrast, are made of quarks and share much more with protons and neutrons than electrons. Moreover, they are made of a quark and antiquark, so they contain antimatter as well.
Laser spectroscopy studies both energy levels, what is sometimes called the principle quantum number of the electron or pion, sometimes given the symbol n, as well as the angular momentum quantum number, which is symbolized as l. The transition from one principle level and angular momentum pair (n,l) to another is observed as excitation at a particular light frequency. Pions that occupy particularly high ones around n=15–16 allow for longer lifetimes.
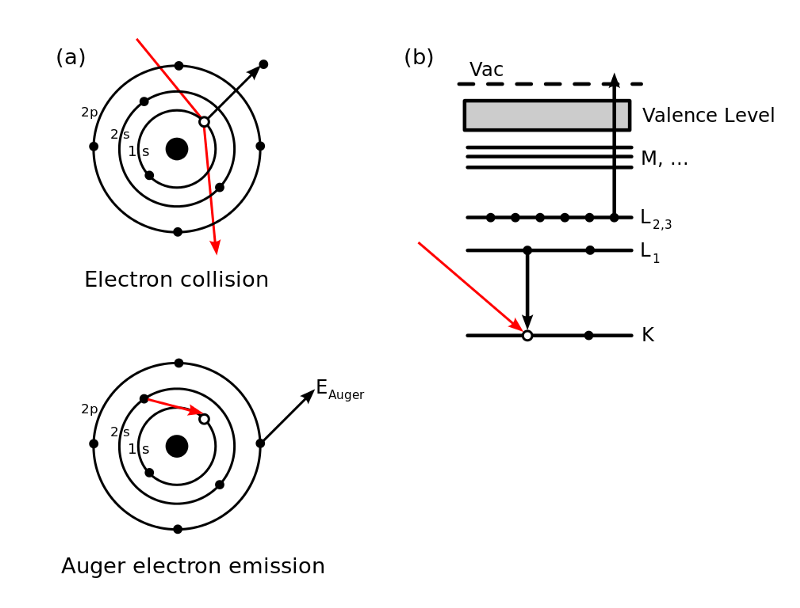
Picosecond lifetime atoms are called “Auger” dominated while those that last for 10s of nanoseconds are called metastable.
The Auger effect, discovered in 1922 by the woman who could be called the mother of the atom bomb, Lise Meitner, refers to the emission of an electron from an atom when an inner vacancy is filled. (It is named after Pierre Victor Auger who independently discovered it in 1923.) Essentially, something hits the atom causing an inner electron to be ejected, another electron fills the hole, releasing its energy, and an outer electron, called an Auger electron, is ejected.
This Auger process, when applied to pions, causes them to rapidly descend into the nucleus where they cause the nucleus to undergo fission. This happens in a trillionth of a second. In the metastable state, however, this does not happen and it takes thousands of times longer for the pions to fall in.
In the metastable state, because the pions are in such a high orbital, the Auger effect is greatly reduced. You can think of this as a spaceship, being in a very high orbit around a planet, takes a while to crash into it (until a laser hits it that is).
Studies of pion atoms could lead to one of the most stringent tests of non-perturbative Quantum Chromodynamics (QCD), the interaction between pions and atomic nuclei via the strong force. Any discrepancies there could lead to new physics. Even now experiments are pushing the boundaries of theoretical calculations and taking QCD to a whole new level of validation.
This level of precision could take physics in new directions entirely since the strong force is far less well understood than electromagnetism. It could even lead to breakthroughs in new, strong or weak force based energy sources. While it may not have the wide press of astrophysics or high energy colliders, exotic atoms are a key component to our overall understanding of the nature of the atom which, like the liar, make up everything.
Hori, Masaki, et al. “Laser spectroscopy of pionic helium atoms.” Nature 581.7806 (2020): 37–41.
Hori, Masaki, Anna Sótér, and Vladimir I. Korobov. “Proposed method for laser spectroscopy of pionic helium atoms to determine the charged-pion mass.” Physical Review A 89.4 (2014): 042515.
Hori, Masaki, et al. “Recent results of laser spectroscopy experiments of pionic helium atoms at PSI.” SciPost Physics Proceedings 5 (2021): 026.
Gotta, D. “Precision spectroscopy of light exotic atoms.” Progress in Particle and Nuclear Physics 52.1 (2004): 133–195.